4 Key Packaging Solutions to Prevent Tampering with Medicine
When it comes to medicine, nothing matters more than safety and trust. For U.S. consumers, taking a prescription or over-the-counter drug should feel safe — and the packaging plays a huge role in giving that reassurance. In this post, we’ll dive into types of packaging that prevent tampering with medicine — or in other words, smart packaging that shows clear signs if someone’s opened or altered the product.
For brands and packaging buyers, it’s not just about “looks” — it’s about real protection, regulatory compliance, and good business practice. We’ll explore four major packaging types, how they work, their benefits, and what you should ask of your packaging supplier.
What “Tamper-Evident” Really Means in Pharmaceuticals
To get started, let’s clarify the language: you’ll often hear tamper-evident packaging, tamper-resistant packaging, and tamper-proof medicine packaging — and they don’t all mean the same thing.
- Tamper-evident packaging for medicine means the packaging has a visible indicator or barrier so that if someone opened it, you’d see signs of tampering. Tamper-resistant implies it’s harder to open, but not necessarily obvious if someone has done so.
- Tamper-proof is a misnomer — no packaging is completely tamper-proof. Experts agree: you need tamper-evident features rather than relying on a single “proof” barrier.
In the U.S. context, brands need to follow regulations like those from the Food and Drug Administration (FDA) and the Drug Supply Chain Security Act (DSCSA) which call for proper tamper-evident and traceability features.
For your brand, using tamper-evident medicine packaging shows consumers you care about safety — not just the pills inside, but the way they arrive and the way they’re stored.
Packaging Type 1: Blister Packs
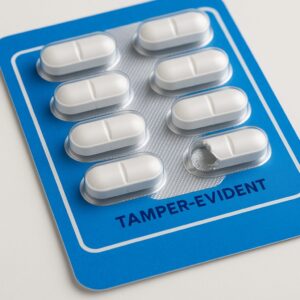
One of the most common and effective solutions: blister packs for medicine.
What they’re and how they work
A blister pack is a formed cavity (usually plastic) sealed with a foil or film backing. Each tablet or capsule is individually packaged. Because each unit is sealed, when you open it or try to tamper with it, it leaves visible signs.
Why it’s effective
- Each dose is unit-of-use packaging, so if one dose appears altered, you know something’s wrong.
- The design itself acts as both a barrier and an indicator.
- For OTC and prescription pills, it’s a clear way to deliver tamper-evident packaging for medicine to U.S. consumers.
Use cases and considerations
Ideal for tablets, capsules, and other solid dosage forms. For U.S. brands choosing blister packs:
- Ensure high-quality foil/backing to prevent moisture/light intrusion.
- Make sure the tamper-evidence is clear to consumers: e.g., “Do not use if blister is opened”.
- As a packaging supplier (like your brand’s packaging line), customise with brand print, dose info, and tamper-evidence messaging.
Packaging Type 2: Induction Seals / Foil Sealed Bottles
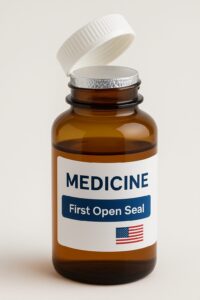
Moving to liquid or multi-dose products: induction seals or foil-sealed bottles.
Description & mechanism
An induction seal is a foil liner that’s heat-sealed to the rim of a bottle under the cap. When you remove the cap or peel off the foil, you visibly break the seal.
How it prevents tampering
Because the foil must be broken to access the contents, if the foil seal is missing or damaged, you know the bottle’s integrity is compromised. Thus, it’s a strong form of tamper-proof medicine packaging (or rather tamper-evident).
Best use cases & design notes
Great for liquid medicines, syrups, jars, multi-dose bottles. U.S. brands should check:
- That the foil is compatible with cold chain or ambient conditions.
- That the seal layer meets regulatory standards (FDA or DSCSA) and clearly indicates first-opening.
- Custom packaging can include printed foil with brand messaging and tamper-evident wording for added trust.
Packaging Type 3: Shrink Bands & Tamper-Evident Caps / Wraps
Now, for another widely used solution: shrink bands and tamper-evident caps or wraps around containers.
What they are
Shrink bands are plastic film bands or sleeves applied over the closure of a container and shrunk to fit tight. Tamper-evident caps may include break-away neckbands or bands that tear away when the cap is opened.
Why they matter
These are particularly visible to consumers: a broken or missing band means “opened”. Therefore, it’s a strong form of tamper-resistant pharmaceutical packaging (again focusing on visible evidence).
Use cases & cautions
- Often found in OTC medicine bottles, supplement bottles, prescription jars.
- When customizing, brands should choose materials that won’t crack or break during shipping and clearly show tampering.
- Consider sustainability: some shrink films may complicate recycling — a newer element to consider in U.S. markets with eco-aware consumers.

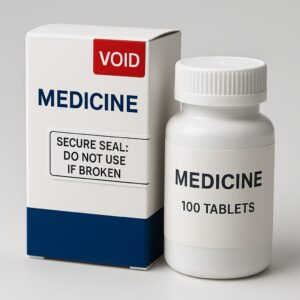
Packaging Type 4: Tamper-Evident Labels/Tapes & Child-Resistant Closures
Finally: tamper-evident labels/tapes and child-resistant closures, which often overlap.
Description & mechanism
Tamper-evident labels or tapes may leave a “VOID” message when removed, or break in a way that cannot be resealed. Child-resistant closures (CRCs) also play a role when combined with tamper-evidence — though their primary aim is safety rather than tamper detection.
When and why you choose this
- Ideal for cartons, folding boxes, mail-order medicine packs, subscription boxes.
- Works well when you already have a bottle but want an additional tamper indicator.
- For U.S. brands: you can emphasise “secure, tamper-evident packaging” as part of your value proposition — especially if targeting conscientious consumers.
Customisation & brand benefit
Your business (IdealCustomBoxes) can offer custom printed tamper-evident labels or tapes, integrating your brand’s design, secure messaging, batch codes etc. This not only protects the medicine but also strengthens brand trust and stands out in the market.
Comparison Table: At a Glance
Here’s a quick comparison to help brands decide which tamper-evident solution fits their needs:
| Packaging Type | Tamper Indicator | Best Use Case | Pros | Things to Consider |
|---|---|---|---|---|
| Blister Packs | Foil/film sealed individual doses | Tablets, capsules | Excellent tamper evidence, unit-dose | Higher cost per unit, less suitable for liquids |
| Induction Seals / Foil Sealed Bottles | Foil liner under cap | Liquids, syrups, multi-dose bottles | Clear first-open signal, strong barrier | Requires proper sealing equipment & compatibility |
| Shrink Bands / Tamper-Evident Caps/Wrappings | Break-away film band around cap | OTC bottles, jars | Highly visible indicator, cost effective | Material choice affects sustainability & recycling |
| Tamper-Evident Labels/Tapes & CRCs | “VOID” labels, security tapes, CRC caps | Cartons, subscription packs, bottles | Customisable branding + tamper indicator | Ensure proper adhesion & consumer ability to open legit pack |
Emerging Trend: Smart & Sustainable Tamper-Evident Packaging
While the four types above form the core of tamper-proof medicine packaging solutions, the best-in-class U.S. brands are already moving further with smart and sustainable packaging.
Smart / Anti-counterfeit
With the rise of anti-counterfeit packaging for pharmaceuticals, features such as unique QR codes, serialization (track & trace), and secure tamper-evident labels are becoming vital. These help brands not only show visible tampering but also verify authenticity in the supply chain.
Sustainable Materials
Consumers care about sustainability — so packaging designers are exploring tamper-evident solutions that use eco-friendly materials. From foil seals with recyclable liners to shrink bands that use less plastic, there’s a trend to balancing custom medicine packaging with tamper-evident features and sustainability.
Why brands should care
In the U.S. market, adopting these trends isn’t just “nice to have” — it enhances brand trust, reduces risk of recalls or counterfeiting, and sets you apart. For packaging buyers, these innovations can be a differentiator in crowded categories.
Checklist for U.S. Medicine Brand Owners / Packaging Buyers
Here’s a handy checklist to use when evaluating packaging options and suppliers:
- Does the packaging provide clearly visible tamper-evidence (bands, foil seals, labels) at first opening?
- Is the packaging aligned with U.S. regulations (FDA, DSCSA) for pharmaceuticals?
- Is the material appropriate for the product (tablet vs liquid, shelf life, moisture/light barrier)?
- Does it enable branding/customisation (print, tapes, labels) while maintaining tamper-evidence?
- For sustainability-conscious consumers: are materials recyclable / low-waste?
- Does the supplier offer custom tamper-evident design options (e.g., custom printed foil seals, “VOID” labels, security tapes)?
- Has the packaging been tested for shipping/distribution environments (vibration, cold chain, handling)?
- Is there compatibility with serialization or anti-counterfeit features if required?
- Internal cost vs risk evaluation: what’s the ROI of investing in premium tamper-evident packaging compared to potential brand or regulatory risk?
- Does the supplier provide documentation/certification for compliance and quality assurance?
Why Choosing the Right Packaging Partner Matters
At the end of the day, even the best tamper-evident design can fail if your packaging partner cuts corners. For U.S. medicine brands looking for custom pharmaceutical packaging solutions that prevent tampering, you should look for:
- Experience with tamper-evident medicine packaging (foil seals, shrink bands, labels)
- Ability to customise for your brand (print, messaging, batch codes)
- Familiarity with regulatory requirements in the U.S.
- Materials and suppliers that meet barrier, shelf-life and sustainability needs
- Transparent pricing, testing, and warranties
By partnering with a fully capable custom packaging provider, you’ll not only meet the functional requirement of tamper-evidence but also use packaging as a brand-asset — reinforcing trust, safety and value in every medicine box or bottle your customers open.
Conclusion
In today’s pharmaceutical marketplace, using types of packaging that prevent tampering with medicine is more than just a nice-to-have — it’s essential. We covered four key packaging types: blister packs, induction/foil sealed bottles, shrink bands/tamper-evident caps, and tamper-evident labels/tapes (with child-resistant closures). We also explored smart and sustainable trends, and provided a practical checklist you can use right away.
If you’re a U.S.-based brand or packaging buyer, it’s time to audit your current packaging strategy: is it clear, visible, trusted, and aligned with best practices? If you need help customising tamper-evident solutions with branding, materials, and compliance built-in — you’re not just investing in packaging — you’re investing in trust.


